For the first time, the US FDA approved a cancer drug for patients based on presence of genetic biomarkers. Whereas cancer drugs have been traditionally been approved based on the location of the tumor, Keytruda (pembrolizumab) is approved for cancer patients with genetic defects classified as “microsatellite instability” or “mismatch repair” deficiencies.
"This is an important first for the cancer community," said Richard Pazdur, acting director of the Office of Hematology and Oncology Products in the FDA's Center for Drug Evaluation and Research and director of the FDA's Oncology Center of Excellence. "Until now, the FDA has approved cancer treatments based on where in the body the cancer started - for example, lung or breast cancers. We have now approved a drug based on a tumor's biomarker without regard to the tumor's original location."
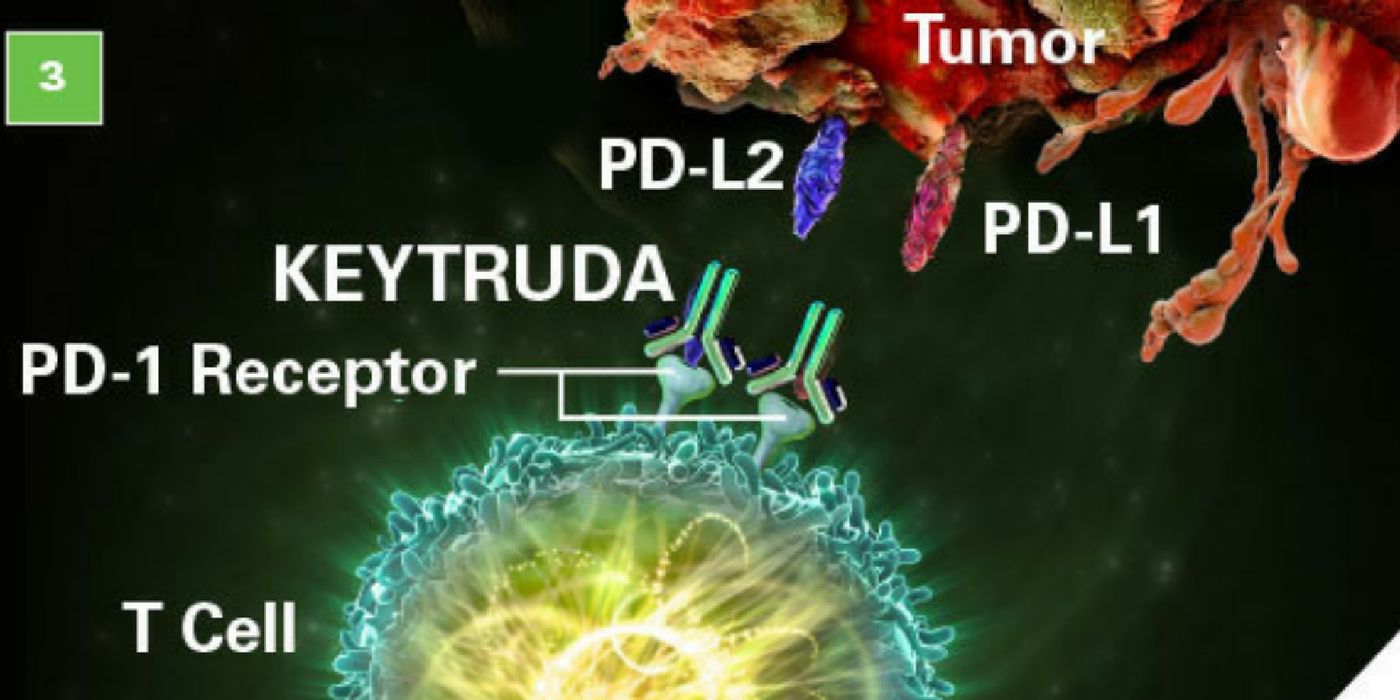
Keytruda is a humanized antibody that works as a second-line immunotherapy agent to destroy the cancer. This drug targets the programmed cell death 1 (PD-1) receptor, a protein that’s responsible for preventing the immune system from attacking its own cells. Cancers can take advantage of this system by making its own proteins that bind to PD-1, which shuts down the body’s ability to kill the cancer. As an immunotherapy, pembrolizumab boosts the body’s ability to kill the cancer again. The drug is already approved for the treatment of several types of cancer, including non-small cell lung cancer, melanoma, and some head and neck cancers, and even mesothelioma.
But the new approval could significantly expand uses of this drug. In particular, Keytruda may now be used to treat any solid tumor as long as the patients have the indicated biomarkers: microsatellite instability-high (MSI-H) or mismatch-repair deficient (dMMR). These types of deficiencies are estimated to be present in about 4 percent of cancers, including colorectal, endometrial, and gastrointestinal cancers.
Notably, the drug’s new indication would expand access to cancer patients with solid tumors that have progressed despite treatment and for which there are no other therapy. For colorectal cancer, this means patients who have exhausted their chemotherapy defenses with drugs such as fluoropyrimidine, oxaliplatin, and irinotecan, Keytruda can be a new option.
“The FDA’s approval of this new indication for Keytruda further supports Merck’s commitment to helping people with difficult-to-treat cancers,” said Roger M. Perlmutter, president of Merck Research Laboratories, the drug’s manufacturer.