Could a long used diabetes drug be repurposed for cancer treatment? Researchers at Rice University have found a potential way to use the diabetes drug class thiazolidinedione to cause breast and ovarian tumor cells to become increasingly susceptible to chemotherapy and the immune system.
Thiazolidinediones are a type of medication that helps modulate fat and glucose metabolism by influencing the upregulation of certain genes and downregulation of others, thereby decreasing insulin resistance in patients with type 2 diabetes. In effect, the drug causes decreased fatty acid circulation causing cells to rely on glucose oxidation for energy.
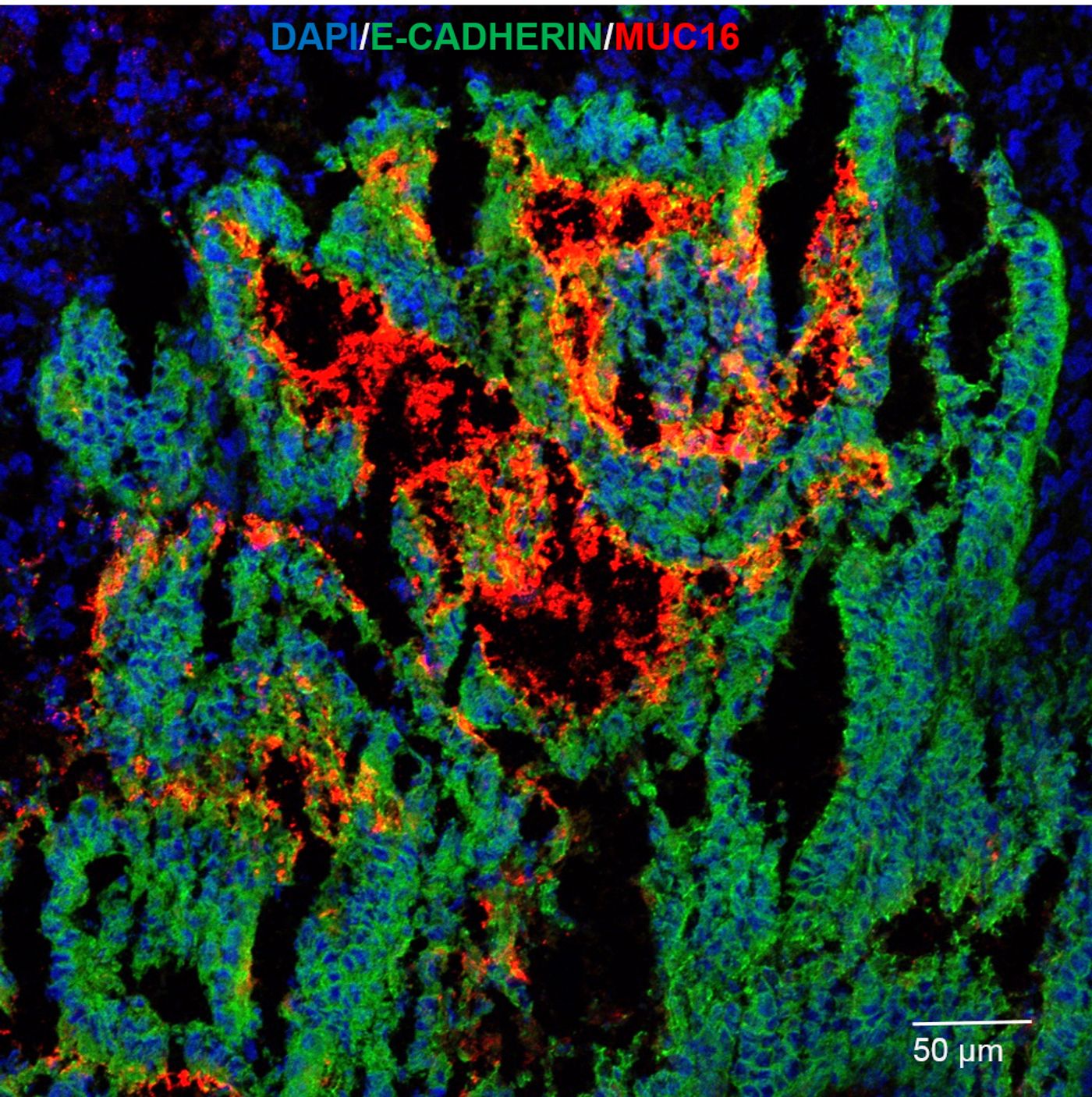
As a side effect, thiazolidinedione can also stop specific glycoprotein expression. These glycoproteins, or mucins, form the protective mucus that lines certain cells and organs in the body such as the stomach, intestines, and lungs. Mucins generally protect cells from exterior exposure. However, cancer cells have enlisted mucins as protection from chemotherapy and attack by the immune system. By covering themselves in mucin proteins, cancer cells create a protective layer around themselves that inhibits immune cells from recognizing a tumor.
In a study published in the
Journal of Cellular Biochemistry, Rice scientist Daniel Carson and researcher Micaela Morgado used the thiazolidinedione drug rosiglitazone to shutdown glycoprotein production in breast cancer cell lines in the lab. Rosiglitazone interferes with cell signaling cytokines that activate a cell’s protective response and increase mucin production. Carson investigated the effects on the mucin MUC16, a glycoprotein that is known to protect breast and ovarian cancer cells.
The idea is by stopping the production of MUC16, the cancer cells can no longer create a protective mucus layer and are more prone to killing by chemotherapeutic agents or our own immune cells.
“If we can intervene in patients and reduce mucin levels, maybe the host immune system can do its job and kill the tumors,” Carson said.
In an interesting twist, their findings presented a conundrum. At low doses, as would be administered to a diabetes patient, the drug actually increased the protective properties of the cancer cells. But at high doses, the drug was able to counteract the protective effect by decreasing the barrier build up enough to allow immune cells to attack. Consequently, in order for the drug to have a positive effect on patients, the drug must be administered at a very high dose.
The Carson lab is learning more about how this interplay functions and is working to adjust drug levels and efficacy. This preliminary study shows great promise for repurposing a currently approved drug to treat a broad range of cancer patients.
“There’s nothing here we can take to a clinic yet,” Carson said. “We learned about ways to intervene and reduce the expression, but we either need better drugs that can do it at much lower, pharmacologically achievable levels, or we need to have a way to target drugs so we can get tumor-specific, superphysiological concentrations that would achieve the goal.
“The upside is that these drugs are already FDA approved,” he said. “If we can repurpose them, we don’t have to go through the same duration of regulatory process required to use them for cancer therapy.”
Sources:
Rice News,
Wiley,
Wikipedia