Surprise! Sugar Molecules Can Form Rigid Structures After all
An oligosaccharide is a small polymer that typically has three to ten individual sugar molecules. Most naturally occurring oligosaccharides can be found in plants. For example, Raffinose, a trisaccharide consisting of galactose, glucose, and fructose, are present in vegetables like beans, cabbage, and brussels sprouts. Oligosaccharides can form conjugates by bonding to amino acid side-chains in proteins or to lipid moieties. Oligosaccharides and their conjugates have many functions in human body. Glycolipids the sugar-coupled fat molecules on the outer cellular membrane can help immune system distinguish the body's own cells from pathogens or other foreign cells. Nutrients wise, oligosaccharides are considered one of the main source of prebiotics. Prebiotics are the broken-down food components that support the growth of certain beneficial bacteria in the colon.
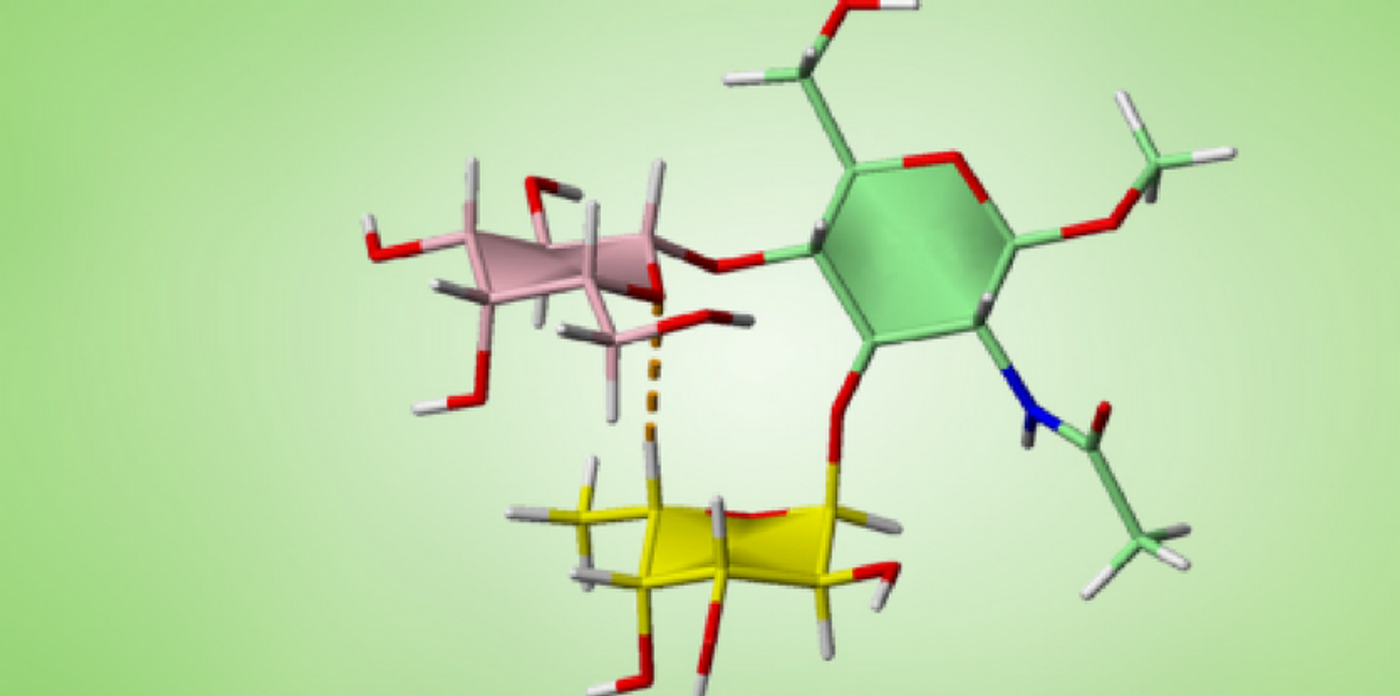
The oligosaccharide that comprises three sugars can form a rigid structure, thanks to a hydrogen bond (dotted line) between Carbon-Hydrogen bond in the upper molecule (red) and Oxygen between two Carbons in the lower molecule (yellow).
Credit: Chemistry
Oligosaccharides were previously thought to be flexible. The individual sugar molecules in the chain are supposed to behave like beads on a string: they can rotate on their position as long as their functional groups do not hinder the motion. Many naturally existing polymers of amino acids or nucleotides cannot do the same - different units within the polymer chain can form hydrogen bond(s), resulting in the so-called secondary structures. These structures are three-dimensional formations, such as the double helix in DNAs or the spirals and pockets in proteins.
Recently the discovery made by an international research team has changed this assumption. Using NMR structure determination and database searches, the researchers identified numerous trisaccharide polymers, all of which contain a fucose, can form a non-conventional C−H⋅⋅⋅O hydrogen bond, which gives these polymers a characteristic rigidity. What is more important, these trisaccharides are the basic building blocks in numerous classes of oligosaccharides found in everything from bacteria to mammals.
This finding disproves a long-standing dogma that the hydrogen bonds between oligosaccharide building blocks are too weak to form any secondary structure. "Our work now shows that certain hydrogen bonds have to be taken into account. As a slight tip of the scales, they can help significantly in forcing the sugar building blocks into a rigid corset," says Dr. Mario Schubert, the head of the research team and a structural biologist at the University of Salzburg. "Oligosaccharides with the newly discovered structural pattern are recognized easily by other molecules using the lock and key principle because a rigid, inflexible structure simplifies molecular recognition." He hopes that their discovery can help to better understand the interactions between oligosaccharides-bearing immune system molecules and stem cells.
Biochemistry of Carbohydrate. Credit: Armando Hasudungan