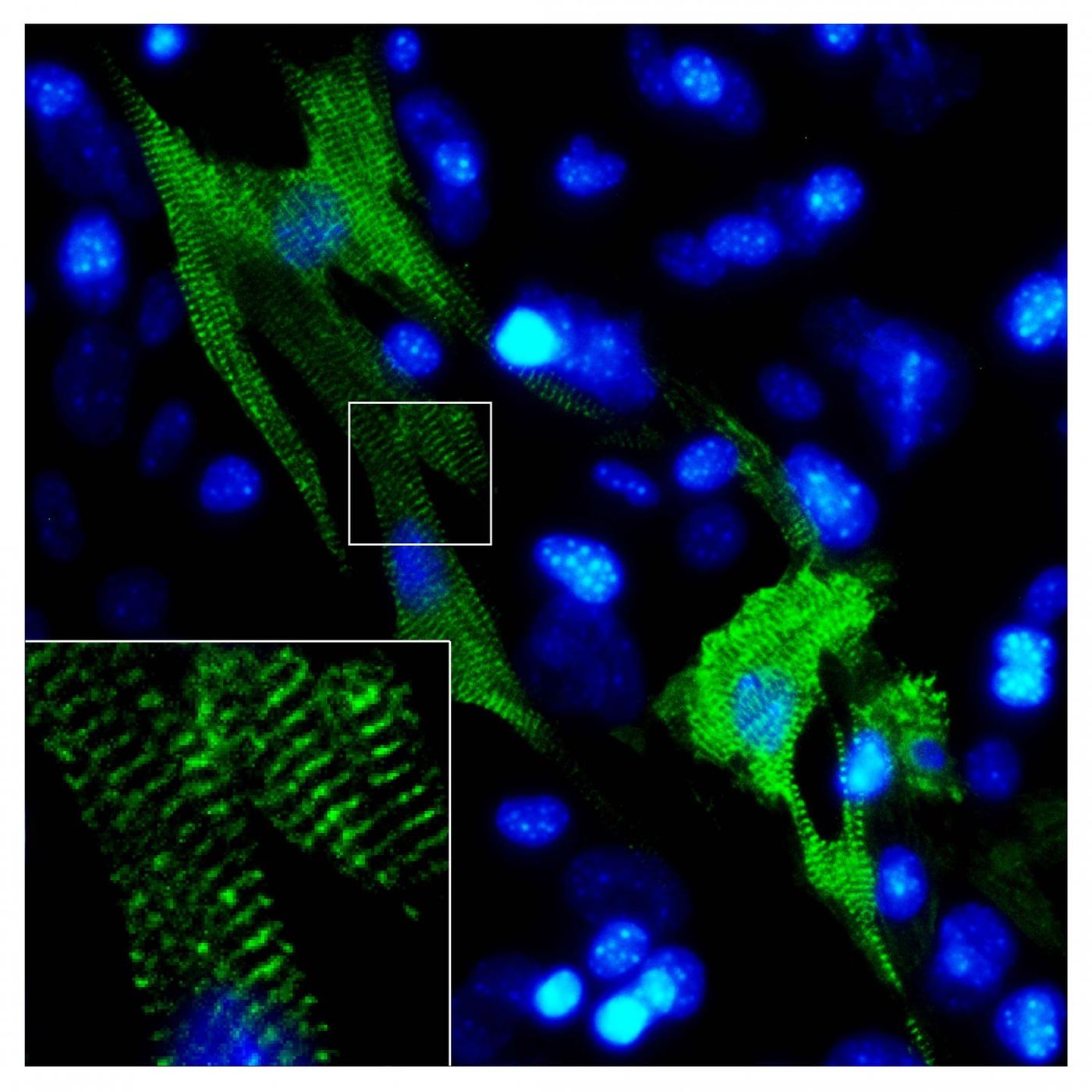
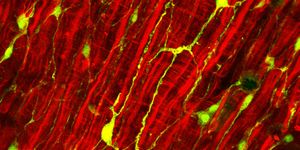
Following a heart attack, healthy heart muscle cells, called cardiomyocytes, die and transform into scar tissue cells, called fibroblasts. Scar tissue doesn’t perform like heart muscle tissue does, and the heart has to work harder to pump the same amount of blood but with fewer cells up for the job. Some organisms are capable of regenerating healthy cells to make up for the loss, but humans are not among them. This inability, paired with insufficient treatment options, is part of what makes heart disease the leading cause of death worldwide.
However, there are two main approaches for manually reprogramming scar tissue cells to become cardiomyocytes again. Which is better? That’s what scientists from the UNC Chapel Hill School of Medicine wanted to know, in the first study to compare the two leading techniques for reprogramming cells and generating patient-specific cardiomyocytes.
The two techniques:
-
Through direct cardiac reprogramming, fibroblasts directly transformed into cardiomyocytes, creating induced cardiomyocytes (iCMs).
-
Through embryonic cell signatures, fibroblasts enter into an embryonic stem cell state, where they are capable of becoming any type of cell. Researchers ensure they become cardiomyocytes, creating induced pluripotent stem cell cardiomyocytes (iPSC-CMs).
Why compare? "Researchers can choose one or the other method based on the specific type of cardiac disease they are interested in studying, while clinicians could carefully select which method is best, considering the pros and cons of each approach,” explained Li Qian, PhD.
Qian and others compared iCMs and iPSC-CMs to both cardiac fibroblasts and natural cardiomyocytes. The two products were largely different in the genes found to be activated or not activated.
"When developing research projects or creating new therapies, we need to know these sorts of genetic features to best help patients,” Qian explained.
iPSC-CMs were more like embryonic cardiomyocytes and iCMs more closely resembled adult cardiomyocytes. iPSC-CMs contained more active genes and more genes ready to be activated or suppressed, but iCMs showed higher expression of genes involved in fatty acid oxidation, “the primary means of energy production in adult hearts.” iCMs also appeared to have greater contractility abilities and were more mature.
“Each method has pros and cons," Qian said. “Our research lays the foundation for understanding the advantages and disadvantages of different approaches for generating patient-specific heart muscle cells for clinical implications, such as disease modeling, drug screening, and ultimately repairing human heart muscle after injuries, such as heart attacks."
The present study was published in the journal Cell Reports.